Biomarker discovery using scRNA-seq: what's the big deal?
Biomarkers have the potential to save lives.
Biomarkers are simply measurable characteristics of organisms, so this may seem like an overstatement. However, the use of biomarkers is becoming increasingly important both in clinical research and practice. Biomarkers for the early detection of disease and the assessment of prognosis are also key components of precision medicine that allow us to find the best treatment for each individual patient. Especially when it comes to complex diseases like cancer.
Recently, there has been a growing interest in using single-cell technologies to identify novel biomarkers. Single-cell RNA sequencing (scRNA-seq) is a powerful tool for identifying gene transcripts within individual cells and can be used to detect changes in gene expression that may be indicative of disease or may predict progression or outcome.
What are biomarkers?
A biomarker can be defined as a characteristic of a therapeutic response or a normal or pathological process that can be objectively and reproducibly assessed and measured. The International Programme on Chemical Safety describes biomarkers as “any substance, structure, or process that can be measured in the body or its products and influence or predict the incidence of outcome or disease”. Simply - biomarkers tell us something about an organism.
There is a wide array of biomarkers. Because of the broad definition, biomarkers include blood pressure and body temperature, as well as more laboratory-measured signs like glucose and viral load. Biomarkers are not a new concept, as medical signs have a long history of use in clinical practice. Currently, biomarkers mostly refer to the most objective, reproducible, and measurable medical signs.
It’s important to note that there are different types of biomarkers (Figure 1). Here, we will focus on prognostic biomarkers to delve deeper into this field!
Figure 1. Types of biomarkers. The figure outlines the most common types of biomarkers and highlights prognostic biomarkers that are emphasized in this article. Various types of biomarkers serve different purposes. Predictive biomarkers provide information on the probability of a response or the lack of it to a particular therapy. Pharmacodynamic biomarkers indicate the molecular and functional effects produced by a drug on a specific target. Safety biomarkers are measured before or after exposure to a drug or an environmental agent to assess its adverse effects. Risk biomarkers are associated with an increased/decreased risk of developing a disease in the case when an individual doesn’t have the clinical disease yet. Monitoring biomarkers are measured repeatedly over time to mainly assess disease progression. Diagnostic biomarkers detect or confirm the presence of a medical condition of interest.
Prognostic biomarkers
Prognostic biomarkers evaluate the risk of an individual developing a specific disease. They can also be used to state the likely course of the disease or the possibility of disease recurrence. Importantly, an accurate prognosis can lead to appropriate clinical support for patients.
This type of biomarker is often used when talking about cancer. It helps to identify tumor-specific characteristics, such as mRNA levels or circulating tumor cells in the blood. For example, some prognostic biomarkers could be used in staging – i.e. to show how far the disease has progressed. This enables doctors to identify the severity of the disease.
A well-known example of prognostic biomarkers used currently is pathogenic germline mutations in the BRCA1 and BRCA2 tumor suppressor genes which indicate an increased risk for breast, fallopian tube, and ovarian cancers in women, and pancreatic and breast cancers in men [1].
The process of getting the biomarker from the discovery phase to clinical practice is difficult. Before a biomarker can be effectively used in the clinic, it has to go through a rigorous validation process that assesses its statistical and analytical performance. For prognostic biomarkers, changes in the value of the biomarker over time irrespective of treatment need to be repeatedly demonstrated.
Even for a biomarker that has been analyzed or has a strong biological basis, the proof of clinical utility may still require additional evidence. It often takes several trials and studies for a biomarker to be completely validated and approved.
Although many biomarkers are already being used in the clinic, there is still a gap when it comes to identifying specific biomarkers for each individual patient and tumor. For example, serum beta-2-microglobulin is used as a prognostic biomarker in various cancer types, but it lacks patient specificity. Biomarkers with high specificity are going to be the cornerstone of future prognostics and precision medicine!
Precision medicine
This article wouldn’t be complete without mentioning the big picture - precision medicine. Precision medicine is advantageous because of its specificity - it’s changing how diseases are identified, classified, and treated. As a result, patients could receive the right treatment at the right time, improving patient care and survival.
Biomarkers are the foundation of successfully achieving precision medicine across a wide range of diseases and conditions. And precision medicine is the reason why biomarkers hold such importance! Through biomarkers, scientists and doctors can make more precise diagnoses, which then leads to more effective and personalized treatments.
Recent advances in science and technology, such as scRNA-seq, have heightened interest in precision medicine. In diseased tissues, single cells can be analyzed to determine what contributes to individual differences between patients, allowing for personalized treatment and the development of biomarkers.
Learn more: Open Precision Medicine Conference
Where does scRNA-seq come in?
Traditionally, biomarkers have been identified using various laboratory techniques, such as immunohistochemistry, ELISA, and PCR. For instance, anaplastic lymphoma kinase translocations which are associated with several cancer types can be identified by RT-PCR or ELISA kits. However, these techniques are often limited in their ability to detect low levels of biomarkers or to identify novel biomarkers.
Single-cell transcriptomics provides unprecedented insight into cellular heterogeneity and changes in dynamic cell populations, as well as molecules and pathways linked to various diseases, and that may be “averaged out” using bulk RNA sequencing.
So, scRNA-seq offers a more focused and comprehensive approach for the identification of new biomarkers. For instance, scRNA-seq provides the opportunity to identify gene expression changes in specific subsets of cells within a tissue that are associated with disease or disease severity.
With the help of scRNA-seq, scientists are already discovering novel biomarkers that could be used in clinical practice or research in the future.
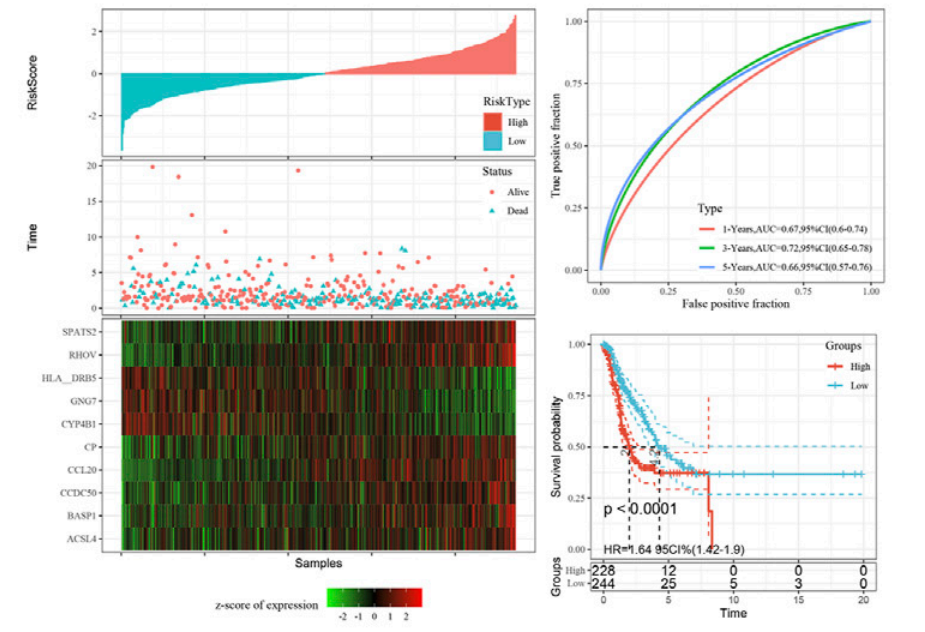
For example, in a recent study, researchers analyzed lung adenocarcinoma single-cell data in combination with bulk RNA-sequencing data and established a novel prognostic risk model for this cancer type [2]. They screened the marker genes of five important cell populations and found 10 genes related to the prognosis of lung adenocarcinoma - CCL20, CP, HLA-DRB5, RHOV, CYP4B1, BASP1, ACSL4, GNG7, CCDC50, and SPATS2 (Figure 2). This 10-gene signature achieved stable prediction efficiency in datasets from different platforms.
Figure 2. The risk score, survival time, survival status, and gene expression in samples based on the 10-gene signature. The risk score reflects the level of risk of disease in the presence of the signature. Analysis of the prognostic classification of the risk score concludes that patients with a higher risk score had a poorer prognosis. Image adapted from Xu et al. (2022).
In another study, autophagy-related 16 like 1 gene (ATG16L1) was associated with poor prognosis in patients with osteosarcoma [3]. So, ATG16L1 is a potential prognostic biomarker in osteosarcoma patients. This gene seems to affect osteosarcoma by mediating CD8+ T cells, especially in patients with metastases.
Since scRNA-seq is a relatively new method, for many diseases, researchers are still in the biomarker discovery phase. In the previously mentioned works, researchers explored cancer single-cell data to find potential prognostic biomarkers. This means that the reliability of these biomarkers still needs to be investigated.
However, there are currently several ongoing clinical trials that aim to discover and validate molecular biomarkers that show reproducible and reliable results using scRNA-seq. For example, in a currently recruiting clinical trial researchers are trying to identify prognostic and diagnostic biomarkers according to the characteristics and evolution of systemic sclerosis and inflammatory myopathies (ClinicalTrials.gov Identifier: NCT04917705).
In the future, these prognostic biomarkers identified by scRNA-seq can allow for the reliable identification of individuals with a more aggressive tumor progression and, thus, a higher chance of disease recurrence and death. As a result, medical practitioners could develop management strategies that ensure these patients receive appropriate and personalized care.
Learn more about other uses of scRNA-seq: Top 5 most revolutionary uses of single-cell RNA sequencing
Biomarker data analysis
Single-cell transcriptomics data can be complex, noisy, and large-scale. This presents challenges in scRNA-seq analysis and thus biomarker discovery. If you are looking for an easier way to explore your data, try out Cellenics® - an open source user-friendly tool for single cell RNA-seq data analysis and visualization!
Researchers already use the Biomage-hosted community instance of Cellenics® to discover new biomarkers by exploring clinical trial data. It’s an open-source and cloud-based scRNA-seq analysis software that allows you to analyze your dataset without prior programming knowledge. It’s fast, user-friendly, and free for academic researchers with datasets of up to 500,000 cells!
References
[1] https://oncologypro.esmo.org/education-library/factsheets-on-biomarkers/brca1-and-brca2-in-ovarian-cancer
[2] Xu, Y., Wang, Y., Liang, L. and Song, N., 2022. Single-cell RNA sequencing analysis to explore immune cell heterogeneity and novel biomarkers for the prognosis of lung adenocarcinoma. Frontiers in Genetics, 13. doi: 10.3389/fgene.2022.975542.
[3] Qin Z, Luo K, Liu Y, Liao S, He J, He M, Xie T, Jiang X, Li B, Liu H, Huang Q, Tang H, Feng W, Zhan X. ATG16L1 is a Potential Prognostic Biomarker and Immune Signature for Osteosarcoma: A Study Based on Bulk RNA and Single-Cell RNA-Sequencing. Int J Gen Med. 2022;15:1033-1045. https://doi.org/10.2147/IJGM.S341879